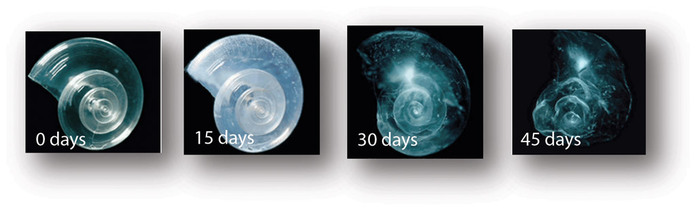
The Effect that Different pH levels has on Calcium Carbonate Shells
Introduction:
The ocean takes up 73% of our beloved earth. People rely so heavily on it for so many different reasons; it is very significant. If it wasn't obvious before, hopefully it is now. Although the ocean deserves to be nurtured and admired, it isn't. The drop of its pH has become a problem to not only humans, but shelled organisms. Since shells are created with calcium carbonate, the acidity level has caused them a bit of trouble. If shells of sea organisms begin to completely dissolve through ocean acidification, these organisms that inhibit the shells will no longer have protection or a form of defense, soon resulting in death. This lab was performed to demonstrate the amount that the acidity affects a calcium carbonate shell.
If pH levels range from 0 to 6, then calcium carbonate shell has a higher chance of dissolving. Since this fact has already been established, the hypothesis of this lab experiment will assume the same thing. If a shell is in a pH level of 0 it will most certainly dissolve faster. Shells that a placed into a pH level that ranges from 7 to 14 will not dissolve, because it will not affect the calcium carbonate. Hypothesis: Shells will bubble up in the vinegar and it will turn more basic when it goes up on the pH scale (7 to 14). Th final mass of the shell will weigh more than the initial mass.
Methods and Materials:
The lab included two beakers containing 150mL of vinegar, while the other had 150mL of saltwater. The vinegar acted as the acid, while the salt water acted as the control. For shells were used: two untreated, one treated with higher acidity, and one treated with lower acidity. The two untreated shells were labeled with an "E" for Experimental, while the other one was labeled with a "C" for control. Before any form of experiment was done, the masses of the two shells were measured and recorded separately. The E shells was then placed in vinegar for 30 minutes, while the C shell was placed in saltwater for 30 minutes. The masses of the low and high concentration shells were measured separately and recorded. Tweezers were used to remove the E shells from the vinegar and the same applied to the C shells. The final masses were recorded and to determine the strength of the shells, a book test was conducted. Stacks of books were piled onto the shells separately until it broke. This would determine how much pressure each shell could handle before finally breaking.
Sample Size: 5 groups of 2 people, 4 shells per group. 2 beakers per group, one full of vinegar and the other salt water.
Control Group: "C" shell labeled for control, placed into a beaker containing 150mL of saltwater.
Experimental Group: "E" shell labeled for Experimental, placed into a beaker containing 150mL of vinegar.
Table 1: Observations of Calcium Carbonate Shells in Vinegar, Saltwater, High and Low Exposures, and its tested strength
Shells Initial Mass Final Mass Mass Difference % Change Weight to Crush
Low 2.810 g 2.880 g -0.07 g -2.49% 3 books
High 5.580 g 5.430 g 0.15 g 2.69% 4 books
E Shell 4.520 g -3.870 g 0.65 g 14.4% 1 book
C Shell 2.810 g 2.880 g -0.07 g 2.49% 3 books
The results in Table 1 suggest that the E shell had the greatest difference which indicates that the acidity did make the shell grow weaker. The weight to crush the shell further proves this because it only took 1 book to crush the shell, versus 4 or 3 books like the high exposure shell or the control shell.
Conclusion:
Based off of the indicated data table, a drop in pH level does have a negative affect on Calcium Carbonate shells. The acidity level causes the shells to weaken and eventually break. This is demonstrated by the amount it took to break the E shell; only 1 book. The estimated weight it took to break a shell is what the C shell was added to the experiment for. By comparing the C shell and E shell, it is easy to see the real effect upon shells in different pH levels. However, the initial mass for the High shell was less than the final mass for some reason. This could have occurred because of a small error in the experiment, maybe a mutation of some such. Although there could have possibly been an error in the E shells, the data does roughly signify that dropping levels of pH can cause harm to calcium carbonate shells.
The Effects that Carbon Dioxide has on Sea Water
Introduction:
Industrialization has many effects on the environment. One being the rise of ocean acidity levels. Tons and tons of carbon is absorbed by the ocean primarily because it is a carbon sink. Statistics show that the levels have increased to about 30% in the last several years. It is important for the waters of the earth to keep a balance between all things, especially for organisms such as plant and animals that inhabit these oceans. Fossil fuels aren't exactly a beneficial help to the environment either. They release carbon dioxide through oil, burning of coal, etc. which results in destruction of the balance that should be maintained. There are three steps to the ocean absorbing carbon dioxide: Absorption of CO2, forms carbonic acid by bonding with water molecules, and separates he carbonic acid into hydrogen bonds. This three step process is what causes the rise of ocean acidity.
Since the balance that should be maintained is important, this lab was designed to demonstrate how far away the acidity levels are from the balance. The effects of carbon dioxide on sea water will be tested and data will be conducted.
Methods and Materials:
Carbon dioxide was placed into both distilled and ocean water. This was accomplished by pouring 100mL of saltwater to a 500 mL beaker, and then adding 1 mL of universal indicator by using a transfer pipette. In order to keep track of the pH color, a paper towel was placed under the beaker. By the time the pH level was recorded, saran wrap was placed over the beaker to form a smooth surface for a straw to be poked in. Some blew into the straw to form bubbles within the liquid of the beaker for two minutes. The color change was recorded within every 30 seconds. A data table and graph were then created. The same steps applied to the room temperature, tap water, ice saltwater and hot saltwater.
Table 1: Data of how the effects of Carbon Dioxide on Sea Water
CONTROL
0 Seconds 30 Seconds 60 Seconds 90 Seconds 120 Seconds
7.8 pH, Dark green 7.3 pH, Green 6.5 pH, Light green 6.4 pH, Light green 6.3 pH,
Light green
COLD
0 Seconds 30 Seconds 60 Seconds 90 Seconds 120 Seconds
7.9 pH, Dark green 7.6 pH, Dark green 7.0 pH, Green 6.5 pH, Light green 6.4 pH,
Light green
Hot
0 Seconds 30 Seconds 60 Seconds 90 Seconds 120 Seconds
8.1 pH, Dark green 7.2 pH, Green 7.0 pH, Green 7.0 pH, Green 6.8 pH,
Light green
Tap
0 Seconds 30 Seconds 60 Seconds 90 Seconds 120 Seconds
7.8 pH, Dark green 6.8 pH, Light green 6.5 pH, Light green 6.4 pH, Light green 6.2 pH, Yellow
Conclusion: According to the graph and data table, blowing air into a straw did create carbon dioxide within the saltwater. To further support this conclusion, the same outcome occurred in the cold, hot and tap water as well. The initial color of the water started off as light green, which signified the neutrality of the pH level. While the bubbles were created by the pressure of carbon dioxide through a narrow straw, the pH level shifted to 6-5 and turned to an even lighter shade of green, almost yellow. These results indicate that different types of water are in fact, affected by carbon dioxide.
